Introduction
Bone contains an outer compact layer, a spongy inner layer, and a medulla comprising the bone marrow. The outer layers are sparsely populated with cells and are basically non-vascular. The only vessels found within bone are periosteal arteries and penetrating nutrient arteries perfusing the bone marrow. As such, mineralized bone tissue exists at relatively low pO2 from 1.7 – 7.2 kPa (12.8 – 54.0 mmHg).
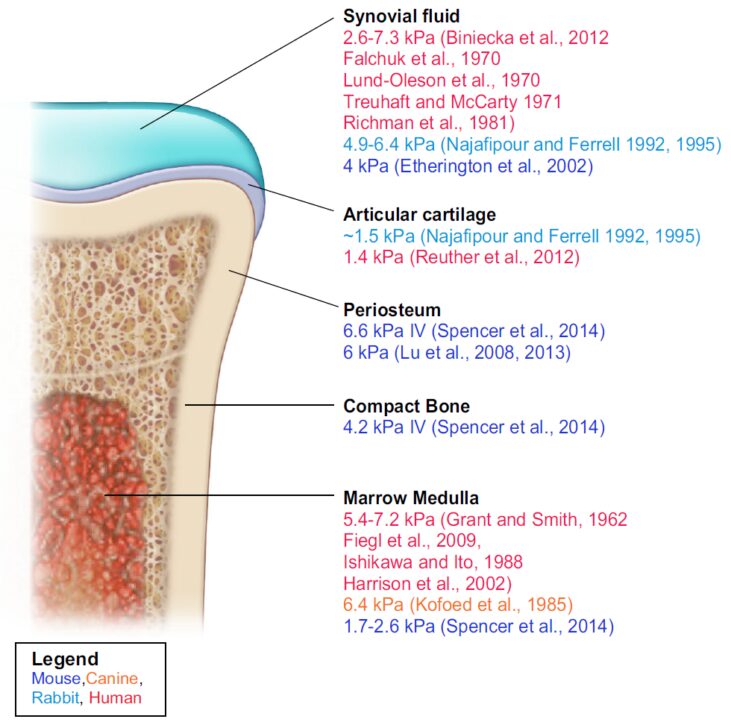
(Above image taken from the 2017 review by Thomas P. Keeley and Giovanni E. Mann, Defining Physiological Normoxia for Improved Translation of Cell Physiology to Animal Models and Humans)
It may seem trivial, but oxygen is vital to numerous cellular processes. Molecular systems have been developed to quickly adapt to oxygen fluctuations, and HIF factors represent their major regulators. HIF, a versatile regulator, has been shown to have various downstream effects and can influence many systems including metabolism, angiogenesis and cell death to name a few.
So, the question remains, how do low oxygen levels (hypoxia) affect osteo-biology? The recent 2020 review by Hannah S. et al. "Take My Bone Away?” Hypoxia and bone: A narrative review delves deeply into this topic. The group looks at data from both cellular and physiological studies to provide a broad, in-depth look into the role of oxygen in bone health. The review itself touches on several main areas and we have attempted to outline some of the more significant points below.
Key definitions
• Osteoclasts are responsible for aged bone resorption
• Osteoblasts are responsible for new bone formation
Hypoxia and Bone
Key points:
• Small changes in either inspired oxygen or oxygen delivery may influence cell homoeostasis, namely by stimulation of HIF pathways.
• Osteoblast bone formation and osteoblast matrix mineralization are both diminished when exposed to low‐oxygen environments.
• Low oxygen has been shown to stimulate increases in osteoclast number and activity.
HIF‐1α and Bone
• First reported that hypoxic/HIF stimulation led to increased bone formation in mice via HIF‐1α‐dependent induction of the angiogenic factor, vascular endothelial growth factor (VEGF).
• HIF‐1α transcription of VEGF improves local vascularisation and, in turn, increases activation of target genes, progenitor cells and nutrients enhancing bone formation.
• It should be noted that the osteoanabolic response is attributed to HIF‐1α stimulation alone and may not prevail in hypoxia, due to the multifaceted nature of reduced oxygen content or delivery.
• Nonetheless, it would seem evident that the hypoxic‐induced increase in osteoclast activity is, in part, HIF‐1α mediated.
HIF‐2 and Bone
• HIF‐2 negatively regulates osteoblast differentiation and bone formation under hypoxia.
• HIF‐2 may inhibit the osteoangiogenic response mediated by HIF‐1, but this remains uncertain.
• Further research is needed to fully understand bone cell crosstalk between HIF-1 and HIF-2 during hypoxia.
Simulating Hypoxia
• Hypoxic mimicking agents such as cobalt or l‐mimosine; gene therapy and iron chelators have been used in studying bone. Interestingly, bone cells appear to respond very differently to each of these stimuli.
• As such, the wide degree of variance stimulating cellular “hypoxia” makes it difficult to ascertain the true bone cell response.
• While not mentioned inherently in the article, the most physiologically relevant way to eliminate this variance and mimic the in vivo state would be to control the O2 microenvironment itself in an oxygen controlled system such as HypoxyLab.
Metabolism, Reactive Oxygen Species, Acidosis on Bone
• Reduced oxygen availability is accompanied by the stimulation of hypoxic pathways, glycolysis, ROS, acidosis.
• It seems plausible that hypoxia stimulates further glucose metabolism, which may be responsible for the increased number and activation of hypoxic osteoclasts.
• Increases in glycolytic metabolism also is linked to positive osteoblast function.
• Hypoxia seems to have decrease differentiation lad lower mineralization in osteoblasts when looking at ROS and acidosis. Alternatively, osteoclasts seem to have a heightened resorption and differentiation under low oxygen conditions.
To conclude
It is important to note that any bone related research being done in vitro on a lab bench or in a standard incubator, is being conducted at levels over 100% higher than what is physiologically pertinent. This is essentially hyperoxic to these specific cells and it is worth considering how this may affect results.
The full article that the above summary is an extract of is linked below and if this topic is of interest to you and your research, please do note that Oxford Optronix offers several options for those studying the physiological and/or metabolic relevance of oxygen, regardless of model.
• For those performing cellular studies, the HypoxyLab is the preeminent leader in hypoxic/physoxic workstations and incubators, offering a fully featured benchtop unit that is the only system to utilizes oxygen partial pressure (pO2) to regulate its internal environment for the most precise all-around oxygen control anywhere or anytime.
• Or for those working in animal models, OxyLite is a system that allows for minimally invasive tissue pO2 measurements of a micro-region in nearly any tissue including bone, using fibre-optic oxygen sensors.
For more details here is a link to the full article by Scott S. Hannah, Sonyia McFadden, Andrea McNeilly and Conor McClean: “Take My Bone Away?” Hypoxia and bone: A narrative review